|
11/5/2023 0 Comments Darker color spectra line
The Fraunhofer lines are typical spectral absorption lines. What are the black lines in an absorption spectrum? The black lines here represent emissions and the color represents the absorption points. We can now also use them to determine certain elemental compositions. 
However, modern spectroscopy has given us the ability to see more of these lines. What is the use of line spectra? What are the dark lines seen in the solar spectrum called?įraunhofer lines, in astronomical spectroscopy, any of the dark (absorption) lines in the spectrum of the Sun or other star, caused by selective absorption of the Sun’s or star’s radiation at specific wavelengths by the various elements existing as gases in its atmosphere. Surface Processes and Ecosystem Changes Through Response Analysis We adopt an addiction model, because for many people, eating even a small amount of an addictive food doesn’t make the craving go away-it makes it worse. Bright Line Eating is very structured and takes a liberating stand against moderation. The four Bright Lines are: Sugar, Flour, Meals, and Quantities. These “fingerprints” can be compared to the previously collected ones of atoms and molecules, and are thus used to identify the atomic and molecular components of stars and planets, which would otherwise be impossible. Spectral lines are often used to identify atoms and molecules. What is the significance of line spectra? In an emission spectra electrons are excited to an excited state by thermal or electrical means and then relax back to a lower state and emit a photon of light at a specific energy, which is seen as at a specific wavelength. There are two types of line spectra, emission and absorption. How are the dark lines of an absorption spectrum created?Īn absorption spectrum occurs when light passes through a cold, dilute gas and atoms in the gas absorb at characteristic frequencies since the re-emitted light is unlikely to be emitted in the same direction as the absorbed photon, this gives rise to dark lines (absence of light) in the spectrum. …or molecules, whereas in an absorption spectrum, portions of a continuous spectrum (light containing all wavelengths) are missing because they have been absorbed by the medium through which the light has passed the missing wavelengths appear as dark lines or gaps. What are the black lines in the absorption spectrum? A spectrum (plural spectra or spectrums) is a condition that is not limited to a specific set of values but can vary, without steps, across a continuum. Hence both are same words with no difference. Spectra is the generally accepted plural form of the word spectrum. 
What is the difference between spectra and spectrum?Īnd spectra is the plural of spectrum. What are bright line spectra?Ī bright line spectrum is created when a beam of light passes through an analyte sample where some wavelengths of the light are absorbed by the atoms in the sample therefore, the electrons in those atoms get to an excited state. Absorption spectra are produced when light from a hot object travels through a cooler, dilute gas. 5, consists of dark lines or bands on top of a continuous spectrum. Finally, an absorption spectrum, like the spectrum of sunlight shown in the bottom of Fig. Which type of spectrum produces dark lines?īasic types of spectra. 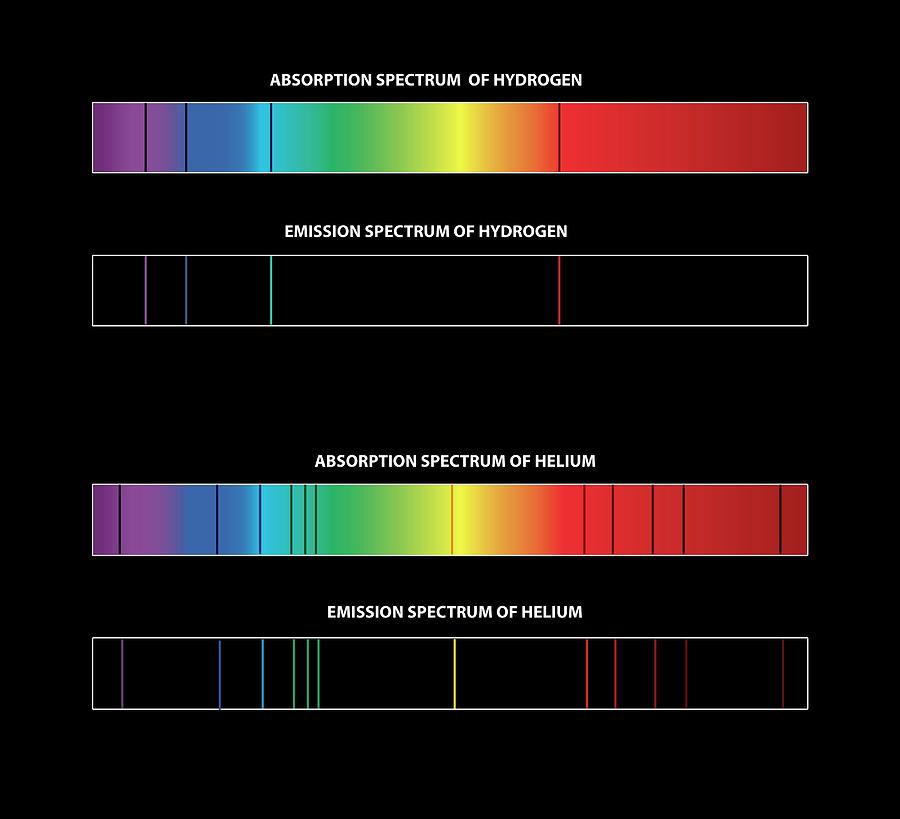
The Fraunhofer lines are any of the dark absorption lines in the spectrum of stars (like the Sun), which is caused by selective absorption of a star’s radiation at specific wavelengths by various gas elements existing in the atmosphere. What causes dark lines in a stars spectrum? What is the difference between a dark line and bright-line spectrum?Ī dark-line, or absorption, spectrum is the reverse of a bright-line spectrum it is produced when white light containing all frequencies passes through a gas not hot enough to be incandescent.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
